Azimuthal TIRF/FRAP/Ablation
Because of its very high signal to noise ratio (SNR) and very low phototoxicity, Total Internal Reflection Fluorescence (TIRF) microscopy is the method of choice to visualize a variety of cellular processes in particular events localized near the plasma membrane of live adherent cells.
Ring / Azimuthal TIRF
However, TIRF classically suffers from interference illumination patterns such as fringes or rings inherent to the technique.
In our theoretical opinion and experience, the best way to generate the nicest TIRF images through a microscope objective is to spin in its back focal plane to average out all artifacts. By using a pair of fast galvanometers, iLas 2 TIRF system is able to spin several times during a single exposure time, even the shortest ones. This speed also allows artefact-free oblique illumination as well as high SNR wide field imaging using the same laser source. To go even further, TIRF/wide-field and multichannel-multi penetration depth acquisition are made possible.
Moreover, its optical design enables to simultaneously combine TIRF with FRAP and ablation.
Classical TIRF
- Single angle
- Fringe interferences
- Heterogeneous fields
Azimuthal TIRF
- Multiple angles
- No fringe interferencies
- Homogeneous illumination (360° rotation angle)
Applications
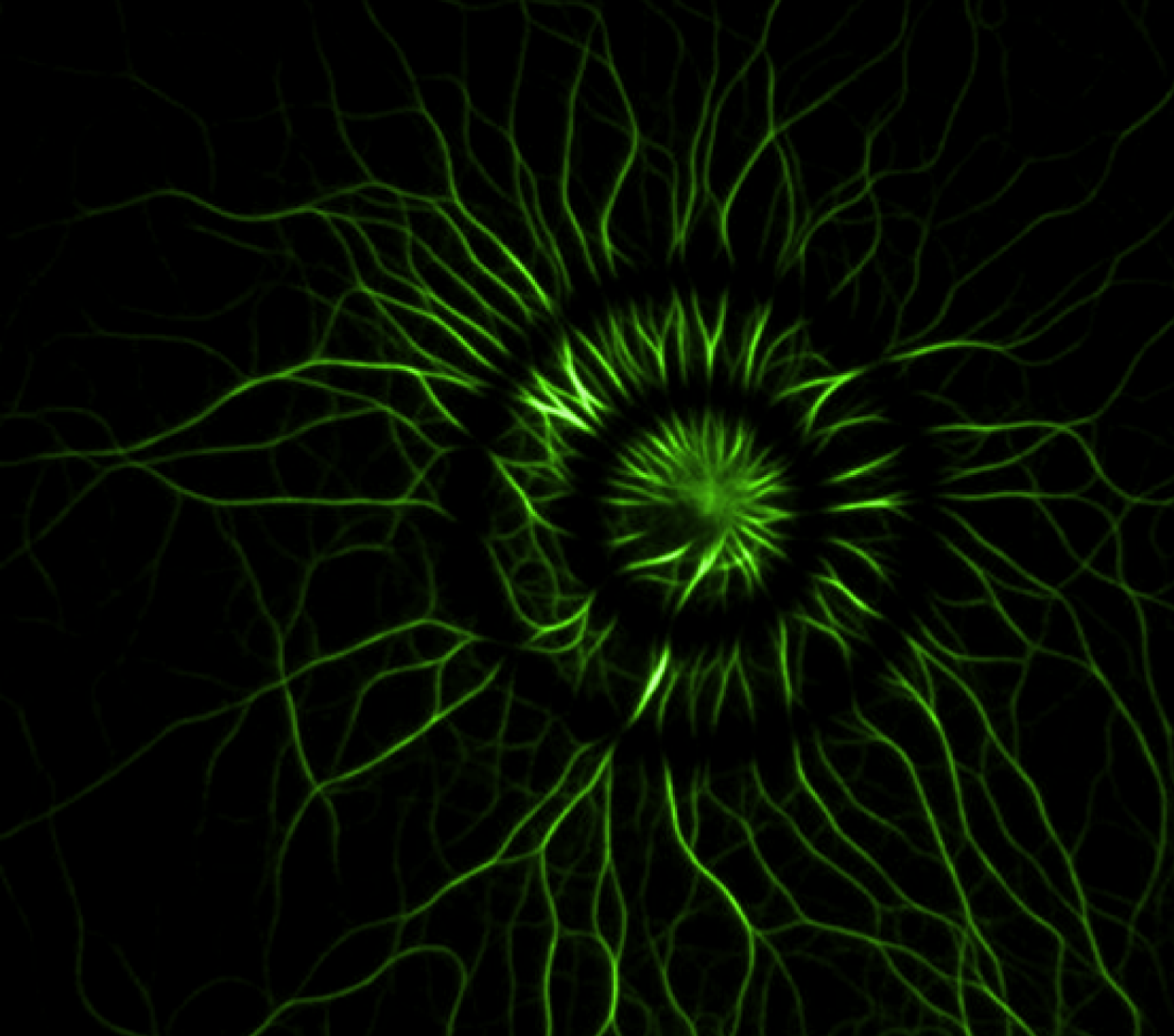
In-vitro actin polymerization
The actin filament growth starts from a longitudinal micro-pattern functionalized with an activator of nucleation. Images were acquired at 1 frame every 10s in TIRF illumination. TIRF is necessary in order to remove the high background of actin monomers in solution. FRAP experiments have been realized to investigate the filaments polarity and growth mechanism from the imposed nucleation geometry.
-

- 0 s
-

- 100 s
-

- 320 s
-

- 370 s
Image courtesy of L. Blanchoin, iRTSV/LPCV, CEA Grenoble.
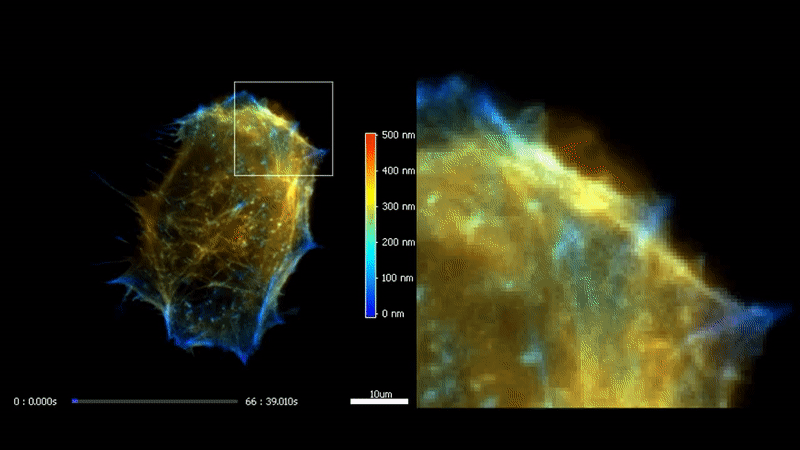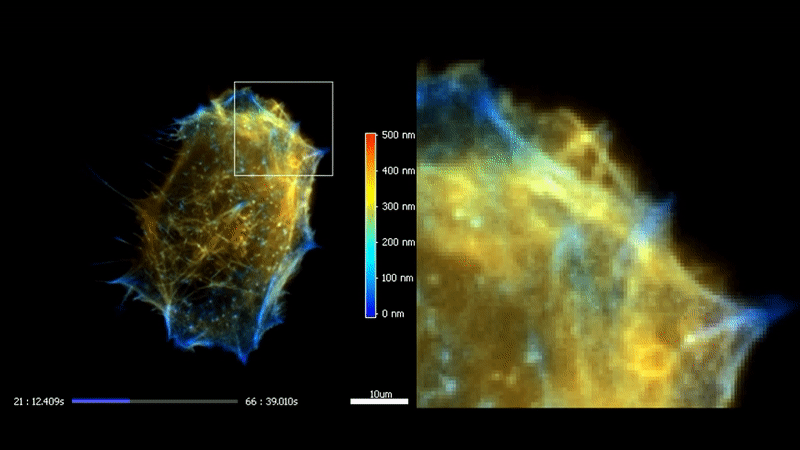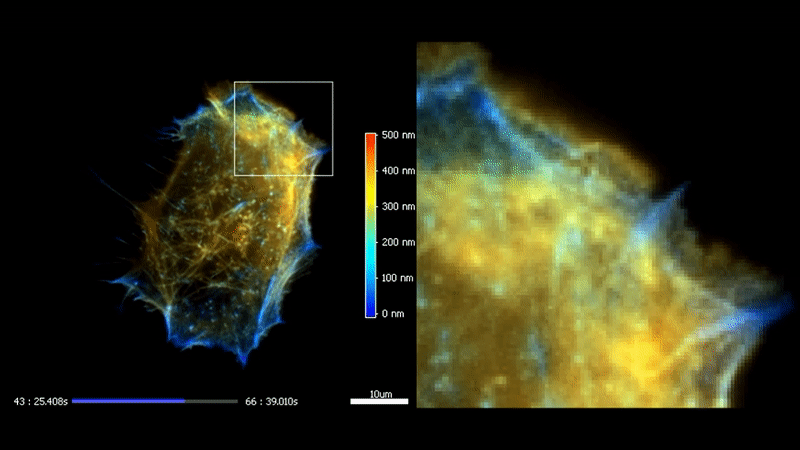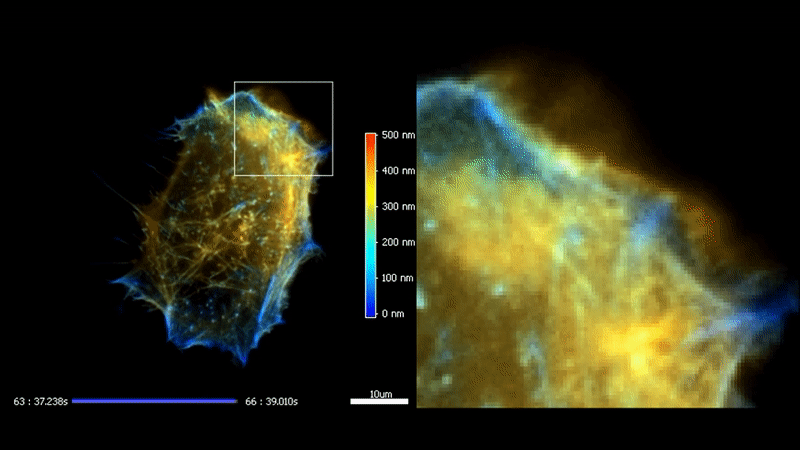
3D TIRF
The high homogeneity, stability and speed of iLas enable to go even futher with TIRF by acquiring a stack of penetration. After processing, it is possible to reconstruct the 3D intensity distribution over a 500 nm range with a 50 nm resolution.
Fast high-resolution 3D total internal reflection fluorescence microscopy by incidence angle scanning and azimuthal averaging. Boulanger et al. PNAS n2014 Dec
Wioland H, Jegou A, Romet-Lemonne G. Torsional stress generated by ADF/cofilin on cross-linked actin filaments boosts their severing. Proc Natl Acad Sci U S A. 2019 Feb
Micro fluidiCS
Proteins of the actin depolymerizing factor (ADF)/cofilin family are the central regulators of actin filament disassembly.
Here, we monitor in TIRF microscopy and quantify the action of ADF/cofilin in different mechanical situations by using single-molecule, single-filament, and filament network techniques, coupled to microfluidics.
The high homogeneity of iLas 2 enables precise quantification of binded proteins along actin filaments.